Home | Facilities | People | Projects | News
Stable Isotopes
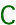 arbon Isotopes
arbon Isotopes
Carbon has two naturally occurring stable isotopes (12C and 13C), and one naturally occurring radioactive isotope (14C). For studies of stable isotope biogeochemistry we measure the ratio of 13C to 12C in samples of interest (e.g., carbonate minerals), expressed relative to the ratio in a standard of known isotopic composition, such as the "Pee Dee Belemnite" (PDB; 13C/12C = 0.00112372 (1) (2)).
The d13C values of terrestrial carbon samples typically range from -40 to +10 per mil (parts per thousand) relative to PDB (3).
The KSC Center for Environmental BioGeoChemistry is not equipped to determine 14C ages.
| Isotope | Atomic mass (ma/u) |
Natural abundance (atom %) |
|---|---|---|
| 12C | 12.000000000* | 98.93 |
| 13C | 13.003354826 | 1.07 |
| 14C | 14.003241982 | half-life of 5715 years
decays by b- to 14N |
See the ![]() page on the
Isotopes of
Carbon.
page on the
Isotopes of
Carbon.
and the ![]() centered on C.
centered on C.
(1)From Table 9-1, p. 154, of David P. Mattey, 1997, Gas source mass spectrometry: isotopic composition of lighter elements, pages 154-170 in Robin Gill (editor), Modern Analytical Geochemistry, Longman (UK).
(2)The uncertainty in this value is much larger than the precision with which we can measure relative ratios, which is one reason for the use the "delta" notation; page 32 in Robert E. Criss, 1999, Principles of Stable Isotope Distribution, Oxford University Press.
(3)From Figure 2-27, p. 111, of Arthur H. Brownlow, 1996, Geochemistry, Prentice-Hall.
website by thorpeallen.net
January 2009
